|
|
|
|
methodology & mechanism Organotin Chemistry Organosilane ChemistryOrganoborane Chemistry total synthesis Please click on the links above for a more detailed research description |
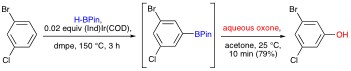
Organoborane Chemistry Like organotins and organosilanes, organoboranes perform beautifully in Pd-mediated cross-coupling reactions also know as Suzuki couplings. Indeed, the Suzuki cross-coupling is the third most common C–C bond forming reaction used for the preparation of drug candidates. Several years ago, we began a collaboration with Professor Milton R. “Mitch” Smith, III to explore catalytic aromatic C-H activation-borylation as a new approach to novel and highly useful aromatic building blocks for organic synthesis. In 1999, Professor Smith and his co-workers first reported on Ir-based catalysts that were remarkably selective for the C-H activation-borylation of unactivated arenes. The mildness of these catalysts and reaction conditions under which they operate allow the borylation of arenes containing a variety of functional groups. Moreover the regiochemical outcomes of these reactions often complement those observed in traditional aromatic substitutions. Owing to these features, catalytic aromatic C-H activation-borylation represents a new and attractive approach to aromatic and heteroaromatic boronic esters. Furthermore, we have designed reaction sequences that merge C-H activation-borylation reactions with cross-couplings, C-N bond forming reactions, oxidations, reductions, deuterium/boron exchange reactions, etc. These processes allow unique access to functionalized aromatic building blocks that are novel or only accessible by protracted, costly, and otherwise unattractive routes. An example of our one-pot C-H activation/borylation/oxidation based preparation of phenols is shown below.
Selected organoborane related publications: “Cobalt-Catalyzed
C-H Borylation of Alkyl Arenes and Heteroarenes
Including the First Selective Borylations of Secondary
Benzylic C–H Bonds” Jayasundara, C. R. K.; Sabasovs, D.;
Staples, R. J.; Oppenheimer, J.; Smith, M. R., III
Maleczka, R. E., Jr. Organometallics 2018,
37, 1567–1574. “Achieving High Ortho Selectivity
in Aniline C-H Borylations by Modifying Boron
Substituents” Smith, M. R., III; Bisht, R.; Haldar, C.;
Pandey, G.; Dannatt, J. E.; Ghaffari, B.; Maleczka, R.
E.; Jr.; Chattopadhyay, B. ACS Catal. 2018,
8, 6216–6223. “Ir-Catalyzed ortho-Borylation of
Phenols Directed by Substrate-Ligand Electrostatic
Interactions: A Combined Experimental / in Silico
Strategy for Optimizing Weak Interactions”
Chattopadhyay, B.; Dannatt, J. E.; Andujar-De Sanctis,
I. L.; Gore, K. A.; Maleczka, R. E.; Jr.; Singleton, D.
A.; Smith, M. R., III J. Am. Chem. Soc. 2017,
139, 7864–7871. “Reversible Borylene Formation from Ring-Opening of Pinacolborane and Other Intermediates Generated From 5-Coordinate Trisboryl Complexes: Implications for Catalytic C–H Borylation” Ghaffari, B.; Vanchura, B. A., II; Chotana, G. A.; Staples, R. J.; Holmes, D.; Maleczka, R. E., Jr.; Smith, M. R., III Organometallics 2015, 34, 4732–4740. “Harnessing C–H Borylation/Deborylation for Selective Deuteration, Synthesis of Boronate Esters, and Late Stage Functionalization” Kallepalli, V. A.; Gore, K. A.; Shi, F.; Sanchez, L.; Chotana, G. A.; Miller, S. L.; Maleczka, R. E., Jr.; Smith, M. R., III J. Org. Chem. 2015, 80, 8341–8353. “Silyl
Phosphorus and Nitrogen Donor Chelates for
Homogeneous Ortho
Borylation Catalysis” Ghaffari, B.; Preshlock, S. M.;
Plattner, D. L.;
Staples, R. J.; Maligres, P. E.; Krska, S. W.; Maleczka,
R. E.; Jr.;
Smith, M. R., III J.
Am. Chem. Soc. 2014, 136,
14345–14348. “A Catalytic Borylation /
Dehalogenation Route to
ortho-Fluoro
Arylboronates” Jayasundara, C. R. K.; Unold, J. M.;
Smith, M. R., III;
Maleczka, R. E.; Jr. Org.
Lett. 2014,
16, 6072–6075. “A Traceless Directing Group for
C–H Borylation”
PPreshlock, S. M.;
Plattner, D. L.; Maligres, P. E.; Krska, S. W.;
Maleczka, R. E.; Jr.;
Smith, M. R., III Angew.
Chem. Int.
Ed. 2013,
42, 12915–
12919. “High Throughput Optimization of
Ir-Catalyzed C–H
Borylation: A
Tutorial for Practical Applications” Preshlock, S. M.;
Ghaffari, B.;
Maligres, P. E.; Krska, S. W.; Maleczka, Robert E.; Jr.;
Smith, M. R.,
III J. Am. Chem. Soc.
2013, 135, 7572–7582. “Outer-Sphere Direction in
Iridium C–H Borylation”
Roosen, P. C.;
Kallepalli, V. A.; Chattopadhyay, B.; Maleczka, R. E.,
Jr.; Singleton,
D. A; Smith, M. R., III J. Am. Chem.
Soc. 2012,
134,
11350–11353. “Practical One-Pot C–H
Activation/Borylation/Oxidation: Preparation of
3-Bromo-5-Methylphenol
on a Multi-Gram Scale” Norberg, A. M.; Smith, M. R.,
III; Maleczka, R.
E., Jr. Synthesis
2011, 857–859. “Electronic Effects in Iridium
C–H Borylations:
Insights From Unencumbered Substrates and Variation of
Boryl Ligand
Substituents” Vanchura, B. A., II; Preshlock, S. M.;
Roosen, P. C.
Kallepalli, V. A.; Staples, R. J.; Maleczka, R. E., Jr.;
Singleton, D.
A.; Smith, M. R., III Chem.
Commun. 2010,
46, 7724–7726. “Divergent Synthesis of
2,3,5-Substituted Thiophenes
by C–H Activation/Borylation/Suzuki Coupling"
Kallepalli, V. A.;
Sanchez, L.; Li, H.; Gesmundo, N. J.; Turton, C. L.;
Maleczka, R. E.,
Jr.; Smith, M. R., III Heterocycles 2010, 80, 1429–1448. “Boc Groups as Protectors and
Directors for
Ir-Catalyzed C–H Borylation of Heterocycles” Kallepalli,
V. A.; Shi,
F.; Paul, S.; Onyeozili, E. N.; Maleczka, R. E., Jr.;
Smith, M. R., III J.
Org. Chem. 2009,
74, 9199–9201. “Getting the Sterics Just Right:
A Five-Coordinate
Iridium Trisboryl Complex that Reacts with C—H Bonds at
Room
Temperature” Chotana, G. A.; Vanchura, B. A., II; Tse,
M. K.; Staples,
R. J.; Maleczka, R. E., Jr.; Smith, M. R., III Chem. Commun. 2009, 5731–5733. “Iridium-Catalyzed Borylation of
Thiophenes:
Versatile, Synthetic Elaboration Founded on Selective
C–H
Functionalization” Chotana, G. A.; Kallepalli, V. A.;
Maleczka, R. E.,
Jr.; Smith, M. R., III Tetrahedron 2008, 64,
6103–6114. “Ir-Catalyzed Functionalization of 2-Substituted Indoles at the 7-Position: Nitrogen-Directed Aromatic Borylation” Paul, S.; Chotana, G. A.; Holmes, D.; Reichle, R. C.; Maleczka, R. E., Jr.; Smith, M. R., III J. Am. Chem. Soc. 2006, 128, 15552–15553. “Aromatic Borylation/Amidation/Oxidation: A Rapid Route to 5-Substituted-3-amidophenols” Shi, F.; Smith, M. R., III; Maleczka, R. E., Jr. Org. Lett. 2006, 8, 1411–1414. “One-Pot Borylation/Amination Reactions: Synthesis of Arylamine Boronate Esters from Halogenated Arenes” Holmes, D.; Chotana, G. A.; Maleczka, R. E., Jr.; Smith, M. R., III Org. Lett. 2006, 8, 1407–1410. “C–H Activation/Borylation/Oxidation: A One-Pot Unified Route To Meta-Substituted Phenols Bearing Ortho/Para-Directing Groups” Maleczka, R. E., Jr.; Shi, F.; Holmes, D.; Smith, M. R., III J. Am. Chem. Soc. 2003, 125, 7792–7793. “Remarkably Selective Iridium Catalysts for the Elaboration of Aromatic C–H Bonds” Cho, J.-Y.; Tse, M. K.; Holmes, D.; Maleczka, R. E., Jr.; Smith, M. R., III Science 2002, 295, 305–308.
|