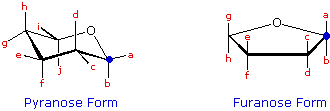
Monosaccharides exist predominantly as cyclic hemiacetals, referred to as pyranose (6-membered ring) and furanose (5-membered ring) forms. For each of seven D-sugars: Arabinose, Ribose, Xylose, Glucose, Galactose, Mannose & Fructose, specify the ring substituents at each of the designated bonds in the following formulas. These substituents will be -H, -OH, -CH2OH, or -CH(OH)CH2OH. Assign a β-configuration at the anomeric carbon ( a blue circle), and keep in mind that these are D-sugars..
Note that the chair conformation specified here may not be the most stable pyranose conformation in every case. It is the configuration at each stereo center that must be correctly specified.
