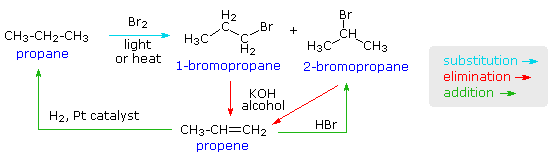
As noted earlier, a chemical reaction is a transformation resulting in a change of composition, constitution and/or configuration of a given compound, referred to as the reactant or substrate. Over the past two hundred year, chemists have observed many millions of reactions involving organic compounds, Understanding the relationship of these reactions to each other provides a context for organizing and remembering them. As a simple example, consider the following transformations of the simple alkane propane.
Bromination, by the action of bromine together with heat or light energy, produces a mixture of 1-bromopropane and 2-bromopropane. We recognize this as a substitution reaction (a bromine atom replaces a hydrogen). Treatment of either bromopropane isomer with an alcohol solution of KOH generates propene by an elimination reaction (H and Br are lost). Propene in turn undergoes addition reactions, such as hydrogen addition to reform propane, and HBr addition to give 2-bromopropane.

In an effort to understand how and why reactions of functional groups take place in the way they do, chemists try to discover just how different molecules and/or ions interact with each other as they come together. To this end, it is important to consider the various components and characteristics of a reaction that may be observed and/or measured as the reaction proceeds. The most common and useful of these are listed below:
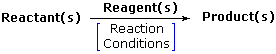
Reactant or Substrate: The organic compound undergoing change in a chemical reaction. Other compounds may also be involved, and common reactive partners (reagents) may be identified. The reactant is often (but not always) the larger and more complex molecule in the reacting system. Most (or all) of the reactant molecular structure is normally incorporated as part of the product molecule.
Reagent: A common partner of the reactant in many chemical reactions. It may be organic or inorganic; small or large; gas, liquid or solid. The portion of a reagent that ends up being incorporated in the product may range from all to very little or none.
Product(s) The final form taken by the major reactant(s) of a reaction.
Reaction Conditions The environmental conditions, such as temperature, pressure, catalysts & solvent, under which a reaction progresses optimally. Catalysts are substances that accelerate the rate ( velocity ) of a chemical reaction without themselves being consumed or appearing as part of the reaction product. Catalysts do not change equilibria positions.
1. Intermediates | |
2. Heat of Reaction | |
3. Reaction Rates |
Since exothermic reactions are energetically (thermodynamically) favored, a careless thinker might conclude that all such reactions will proceed spontaneously to their products. Were this true, no life would exist on Earth, because the numerous carbon compounds that are present in and essential to all living organisms would spontaneously combust in the presence of oxygen to give carbon dioxide-a more stable carbon compound. The combustion of methane, for example, does not occur spontaneously, but requires an initiating energy in the form of a spark or flame. The flaw in this careless reasoning is that we have focused only on the initial (reactant) and final (product) states of reactions. To understand why some reactions occur readily (almost spontaneously), whereas other reactions are slow, even to the point of being unobservable, we need to consider the intermediate stages of reactions.
| Exothermic Single Step Reaction | Endothermic Single Step Reaction | Exothermic Two Step Reaction |
|---|---|---|
 |
 |
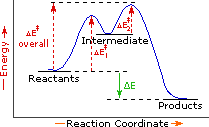 |
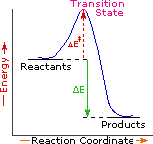
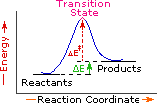
Every reaction in which bonds are broken will have a high energy transition state that must be reached before products can form. In order for the reactants to reach this transition state, energy must be supplied and reactant molecules must orient themselves in a suitable fashion. The energy needed to raise the reactants to the transition state energy level is called the activation energy, ΔE‡.
An example of a single-step exothermic reaction profile is shown on the left above, and a similar single-step profile for an endothermic reaction is in the center. The activation energy is drawn in red in each case, and the overall energy change (ΔE) is in green.
The profile becomes more complex when a multi-step reaction path is described. An example of a two-step reaction proceeding by way of a high energy intermediate is shown on the right above. Here there are two transition states, each with its own activation energy. The overall activation energy is the difference in energy between the reactant state and the highest energy transition state. We see now why the rate of a reaction may not correlate with its overall energy change. In the exothermic diagram on the left, a significant activation energy must be provided to initiate the reaction. Since the reaction is strongly exothermic, it will probably generate enough heat to keep going as long as reactants remain. The endothermic reaction in the center has a similar activation energy, but this will have to be supplied continuously for the reaction to proceed to completion.
1. Temperature Effect on Reaction Rate
What is the source of the activation energy that enables a chemical reaction to occur? Often it is heat in the form of a flame or spark, such as that provided in an internal combustion engine burning gasoline. At room temperature, indeed at any temperature above absolute zero, the molecules of a compound have a total energy that is a combination of translational (kinetic) energy, internal vibrational and rotational energies, as well as electronic and nuclear energies. The temperature of a system is a measure of the average kinetic energy of all the atoms and molecules present in the system. As shown in the following diagram, the average kinetic energy increases and the distribution of energies broadens as the temperature is raised from T1 to T2. Portions of this thermal or kinetic energy provide the activation energy for many reactions, the concentration of suitably activated reactant molecules increasing with temperature, e.g. orange area for T1 and yellow plus orange for T2. (Note that the area under a curve or a part of a curve is proportional to the number of molecules represented.)
| Distribution of Molecular Kinetic Energy at Two Different Temperatures, T1 & T2 | 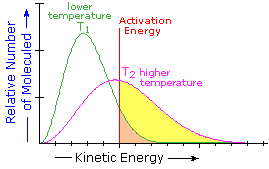 |
|---|
2. Reaction Rates and Kinetics
Chemical reactivity is a primary concern of chemistry, and the study of reaction rates provides essential information about this subject. Some reactions proceed so rapidly they seem to be instantaneous, whereas other reactions are so slow they are nearly unobservable. Most of the reactions described in this text take place in from 0.2 to 12 hours at 25 ºC. Temperature is important, since fast reactions may be slowed or stopped by cooling, and slow reactions are accelerated by heating. When a reaction occurs between two reactant species, it proceeds faster at higher concentrations of the reactants. These facts lead to the following general analysis of reaction rates.
Reaction Rate = |
| • |
| • |
|
|---|
Since reacting molecules must collide to interact, and the necessary activation energy must come from the kinetic energy of the colloding molecules, the first two factors are obvious. The third (probability) factor incorporates the orientational requirements of the reaction. For example, the addition of bromine to a double bond at the end of a six-carbon chain (1-hexene) could only occur if the colliding molecules came together in a way that allowed the bromine molecule to interact with the pi-electrons of the double bond.
The collision frequency of reactant molecules will be proportional to their concentration in the reaction system. This aspect of a reaction rate may be incorporated in a rate equation, which may take several forms depending on the number of reactants. Three general examples are presented in the following table.
Reaction Type | Rate Equation | Reaction Order |
|---|---|---|
A ——> B | Reaction Rate = k•[A] | First Order Reaction (no collision needed) |
A + B ——> C + D | Reaction Rate = k•[A]•[B] | Second Order Reaction |
A + A ——> D | Reaction Rate = k•[A]2 | Second Order Reaction |
These rate equations take the form Reaction Rate = k•[X]n•[Y]m, where the proportionality constant k reflects the unique characteristics of a specific reaction, and is called the rate constant. The concentrations of reactants X and Y are [X] and [Y] respectively, and n & m are exponential numbers used to fit the rate equation to the experimental data. The sum n + m is termed the kinetic order of a reaction. The first example is a simple first order process. The next two examples are second order reactions, since n + m = 2. The kinetic order of a reaction is usually used to determine its molecularity.
In writing a rate equation we have disconnected the collision frequency term from the activation energy and probability factors defined above, which are necessarily incorporated in the rate constant k. This is demonstrated by the following equation.
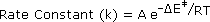
The complex parameter A incorporates the probability factor. Because of the exponential relationship of k and the activation energy, small changes in ΔE‡ will cause relatively large changes in reaction rate. An increase in temperature clearly acts to increase k, but of greater importance is the increase in average molecular kinetic energy such an increase produces. This was illustrated in a previous diagram, increase in temperature from T1 to T2 producing a larger proportion of reactant molecules having energies equal or greater than the activation energy (designated by the red line).
As an approximation for many reactions taking place near room temperature, chemists estimate that the rate of reaction roughly doubles for every 10 ºC increase in the temperature.
1. Reactants and Reagents
A. Reactant Structure: Variations in the structure of the reactant may have a marked influence on the course of a reaction, even though the functional group is unchanged. Thus, reaction of 1-bromopropane with sodium cyanide proceeds smoothly to yield butanenitrile, whereas 1-bromo-2,2-dimethylpropane fails to give any product and is recovered unchanged. In contrast, both alkyl bromides form Grignard reagents (RMgBr) on reaction with magnesium.
| ||
B. Reagent Characteristics: Apparently minor changes in a reagent may lead to a significant change in the course of a reaction. For example, 2-bromopropane gives a substitution reaction with sodium methylthiolate, but undergoes predominant elimination on treatment with sodium methoxide.
|
2. Solvent
|
Most organic reactions take place in solution, and selection of a proper solvent is critical to the success of the reaction. Although water is often referred to as the universal solvent. hydrocarbons and many other organic compounds are relatively insoluble in it. This difficulty is overcome by using hydrocarbon, chlorocarbon, and ethers, as well as ketone and ester solvents, in which organic compounds have greater solubility. When choosing a solvent for a reaction, it is important to avoid solvents having functional groups that might compete with or interfere with the desired reaction. |
3. Catalysts
|
As noted above, catalysts are substances that accelerate the rate of a chemical reaction without themselves being consumed or appearing as part of the reaction product. Catalysts may be soluble (homogeneous) or insoluble (heterogeneous) in the reaction medium. Metals and organometallic compounds often function as catalysts, examples of which in specific reactions will be described when appropriate. The most common type of catalysis encountered in organic reactions is effected by acids and bases. |
When unsymmetrical reactants are combined with unsymmetrical reagents we might expect mixtures of products, representing all possible modes of reaction. In practice, however, we often find a surprising selectivity, favoring one type of product over all others. Results of this kind reflect the fact that competing reactions, even very similar ones, generally have different rates. As expected then, the fastest reaction will produce the dominant product. The experience of many years of chemical experimentation has demonstrated several kinds of such selectivity.
1. Regioselectivity
It is often the case that addition and elimination reactions may, in principle, proceed to more than one constitutionally isomeric product. Thus 1-butene might add HBr to give either 1-bromobutane or 2-bromobutane, depending on which carbon of the double bond receives the hydrogen and which the bromine. In such situations, if one of two (or more) possible products is formed preferentially, the reaction is said to be regioselective. The following equation demonstrates this outcome.
The base induced elimination of 2-bromobutane displays a similar regioselectivity in that a mixture of 2-butene isomers is formed preferentially over 1-butene. Simple substitution reactions do not normally exhibit regioselectivity, since by definition only one constitutional product is possible. However, rearrangements are known to occur during some reactions of this kind. |
2. Stereoselectivity
|
If the reaction products are such that stereoisomers may be formed, a reaction that yields one stereoisomer preferentially is said to be stereoselective. In the addition of bromine to cyclohexene, for example, cis and trans-1,2-dibromocyclohexane are both possible products of the addition. Since the trans-isomer is the only isolated product, this reaction is stereoselective.
|
3. Stereospecificity
|
This term is applied to cases in which stereoisomeric reactants behave differently in a given reaction. Examples include:
B. Different rates of reaction, as in the base-induced eleimination of cis & trans-4-tert-butylcyclohexyl bromide (equation 1 below). C. Different reaction paths leading to different products, as in the base-induced elimination of cis & trans-2-methylcyclohexyl bromide (equation 2 below).
The mechanisms of these substitution and elimination reactions are discussed in the alkyl halide section of this text. |
In view of the subtle differences in the rates and resulting selectivity of many reactions, as noted above, it is helpful to identify some general factors that have a significant influence on all reactions of organic compounds. Some of the most important of these have already been noted, but deserve to be listed here.
A. Electronic Effects: The distribution of electrons at sites of reaction (functional groups) is a particularly important factor. Electron deficient species or groups, which may or may not be positively charged, are attracted to electron rich species or groups, which may or may not be negatively charged. We refer to these species as electrophiles & nucleophiles respectively. In general, opposites attract and like repel. | |
B. Steric Effects: Atoms occupy space. When they are crowded together, van der Waals repulsions produce an unfavorable steric hindrance. Steric hindrance may influence conformational equilibria, as well as destabilizing transition states of reactions. | |
C. Solvent Effects: Most reactions are conducted in solution, not in a gaseous state. The solvent selected for a given reaction may exzert a strong influence on its course. Remember, solvents are chemicals, and most undergo chemical reaction under the right conditions. |
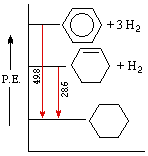 |
An important distinction can now be made between thermodynamic stability and chemical stability. Thermodynamic stability refers to the potential energy of a compound, and is related to the bond energies of its constituent atoms. Chemical stability refers to a compound's resistance to chemical reaction with a variety of reagents, and is related to the activation energy barrier it presents to possible chemical change. A comparison of cyclohexene and benzene provides a good example of this distinction. Both these compounds add hydrogen in exothermic addition reactions that give cyclohexane as a common product. From heats of hydrogenation we find that cyclohexene has a potential energy (P.E.) roughly 28.6 kcal/mol higher than cyclohexane, whereas benzene is 48.9 kcal/mol above cyclohexane. This relationship is graphed on the right. Heats of reaction are commonly given as ΔHº values, where H is a thermodynamic variable called enthalpy or heat content. By convention, exothermic reactions have a negative ΔHº .
We conclude from these measurements that both cyclohexene and benzene are thermodynamically less stable than cyclohexane, and that benzene is thermodynamically less stable than cyclohexene. We know, however, that the chemical reactivity of these unsaturated compounds does not reflect this stability order. Cyclohexene reacts rapidly with bromine, as well as with potassium permanganate and sulfuric acid, whereas benzene is relatively inert to all three reagents (in the absence of catalysts and/or heat). The chemical reactivity of benzene is therefore less than the reactivity of cyclohexene, and we may say that benzene is chemically more stable than cyclohexene - at least toward the reagents noted here.
Just as a chain is no stronger than its weakest link, a molecule may be rendered chemically unstable by one weak bond. We see this in the chemical behavior of peroxides (R–O–O–R). The O–O bond is less than half as strong as a C–C bond, and peroxides are notoriously unstable, decomposing via alkoxy radicals (R–O·) on mild heating. The useful organization of organic reactions by functional groups is a further example of how a few susceptible bonds in a molecule can determine its general chemical reactivity.
| Return to Table of Contents |
|---|
![]()