Welcome to the Jackson Research Group website!
Probing mechanisms and theory from molecular interactions to process design, Jackson group efforts range from fundamental...Nature, scope, and applications of hydridic-to-protonic hydrogen bonding, complexant design and synthesis for thermally robust alkalides and electrides, approaches to organic-based magnetic materials by self-assembly, "Green" catalytic pathways from renewables to useful “petro-” chemicals
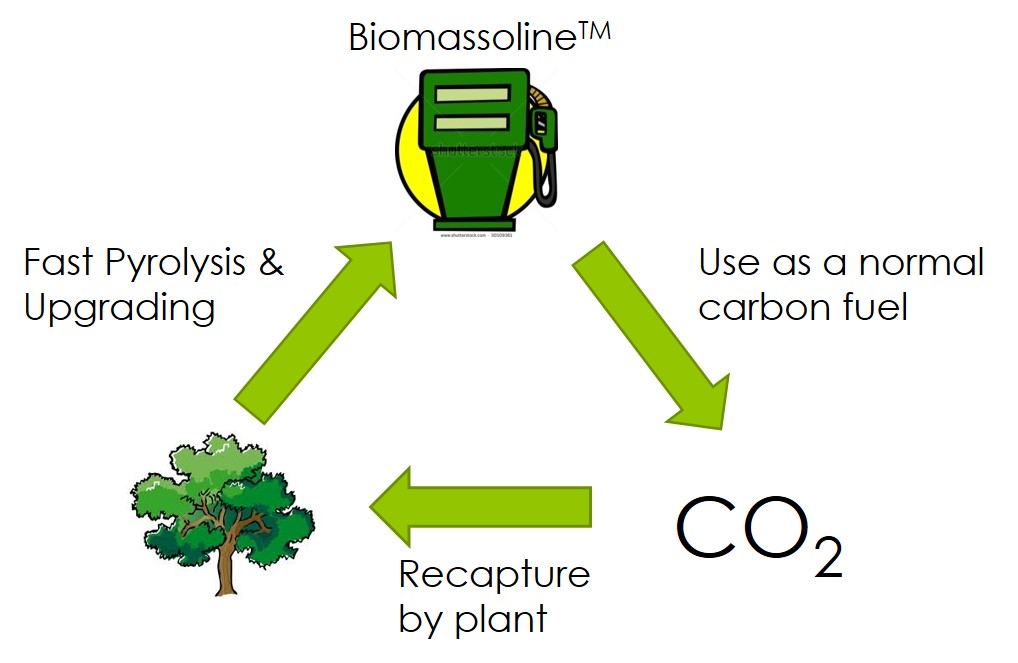
Considering impending petroleum shortages, the environmental costs of using fossil-based fuels, the limitations of the hydrogen-based approach for transport, and the irreplaceable nature and ever growing demand for hydrocarbons, there is a need to shift hydrocarbon production in the US, and ultimately worldwide, to a renewable source: Biomass.....Read More »
Hydridic Protonic Hydrogen Bonding
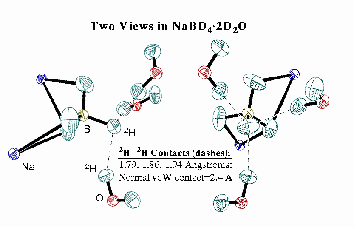
In work started in summer of 1994, and primarily carried forward by summer high school students and undergraduates, we demonstrated an interaction between the electron pair of a B-H bond and a traditional H-X (X=N, O, Halogen) hydrogen bonding partner. Single crystal X-ray and neutron diffraction, solution spectroscopies (IR, NMR), and ab initio calculations all indicate...
Read More »
Lignin Dimer Synthesis and Model Study
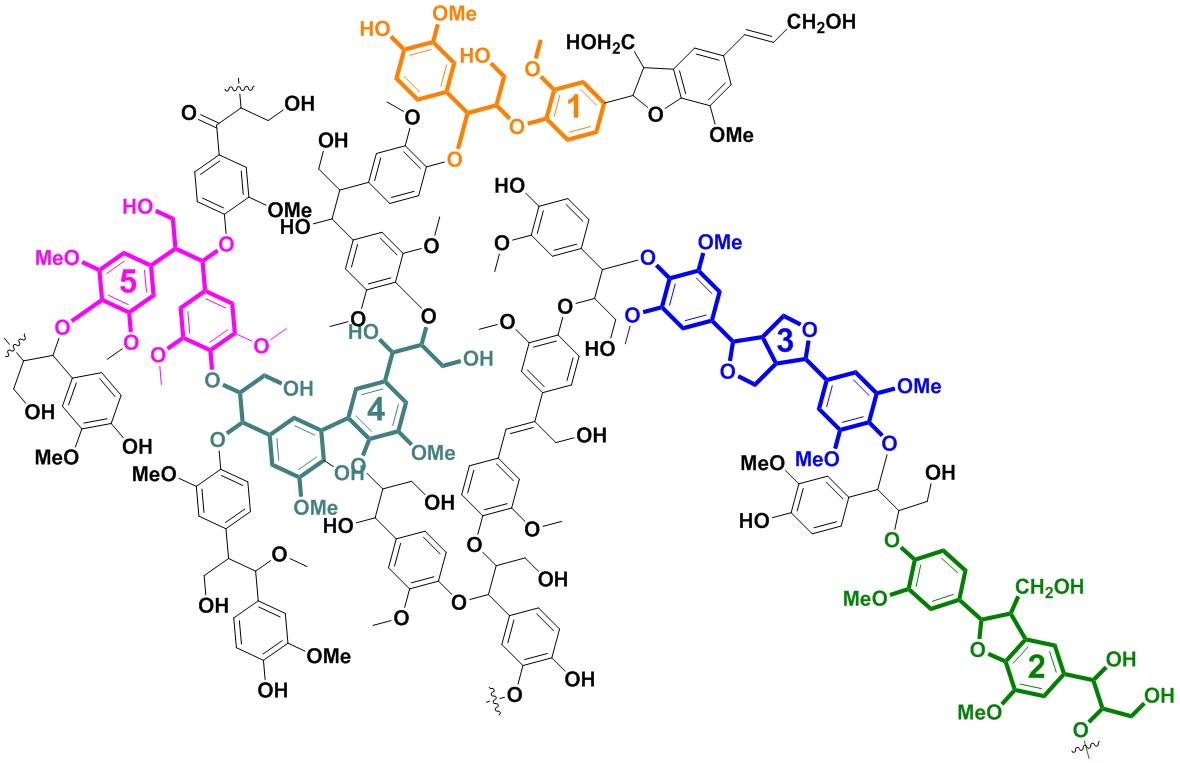
ECH can upgrade the bio-oil produced by pyrolysis of lignin by cleaving the linkages between lignin monounits, thus lowering down the viscosity, stabilizing bio-oil, and increase the energy density of bio-oil. More specifically, model molecules of lignin dimer with β-5, 5-5, β-β linkages....
Read More »
Perazacryptand as a Host for Hydride
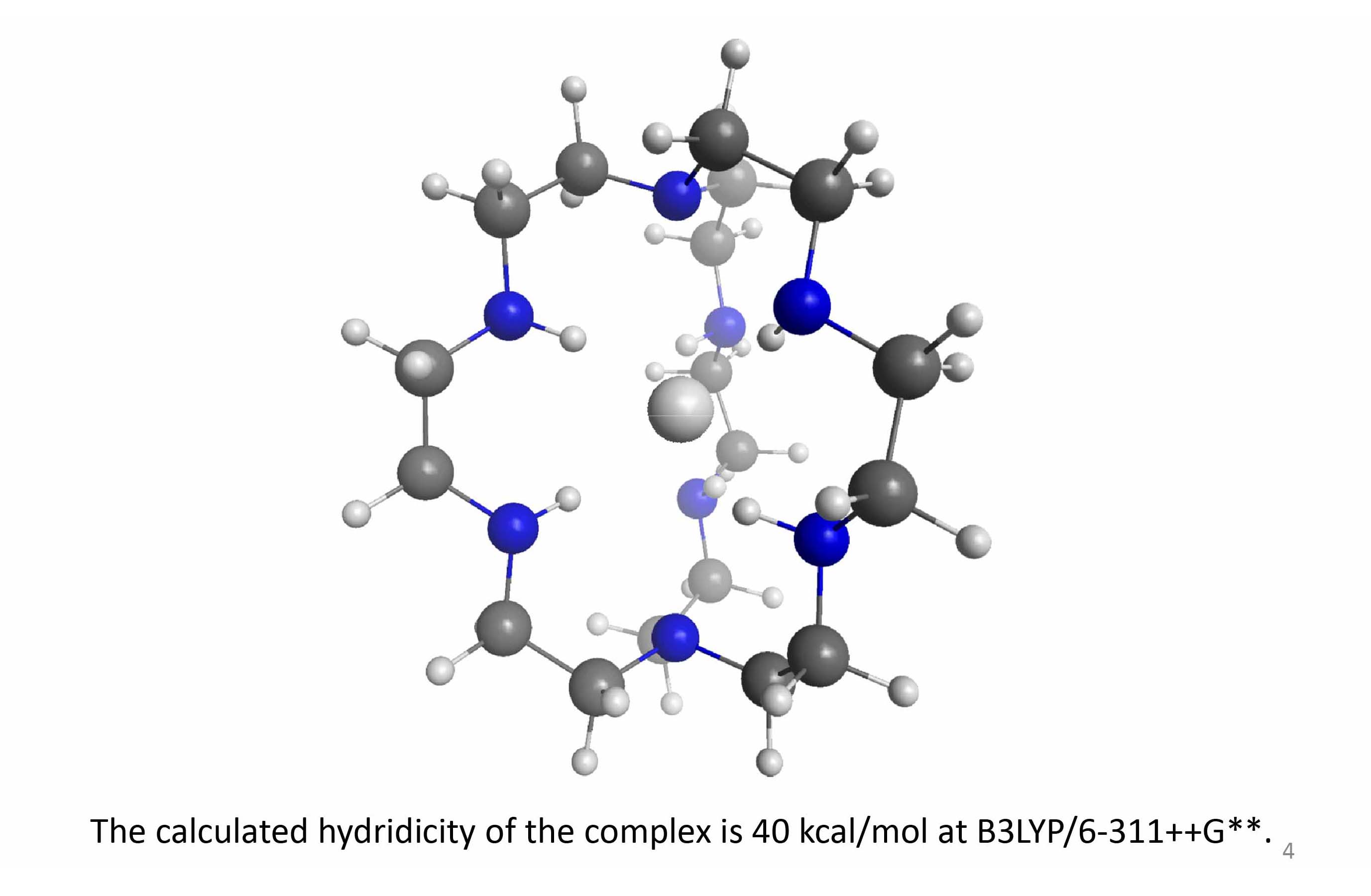
Amine N-H bonds are good candidates for hydrogen bonding with basic anions. In general, the complexing capacity of a moiety X-H in a hydrogen bond X-H…Y- (X, Y = electronegative atoms or groups) increases with the acidity of X-H, or more precisely, with the stability of the X- anion. However, for basic Y- partners....Read More »