Research
Synopsis
The major emphasis of our research is on chemistry and biology of carbohydrates. Carbohydrates play important roles in many biological processes such as inflammation, tumor metastasis, bacterial and viral infections. Building on our strength in synthetic chemistry, we take a multi-disciplinary approach to study the functions and applications of these fascinating molecules. Our research encompasses several areas including synthetic organic chemistry, nanoscience and chemical immunology. Students and postdocs in our group are exposed to a wide variety of state-of-the-art research topics including organic synthesis, physical organic chemistry, nanotechnology, materials science, immunology, cancer biology and molecular imaging. This is an exciting scientific journey!
Synthetic organic chemistry
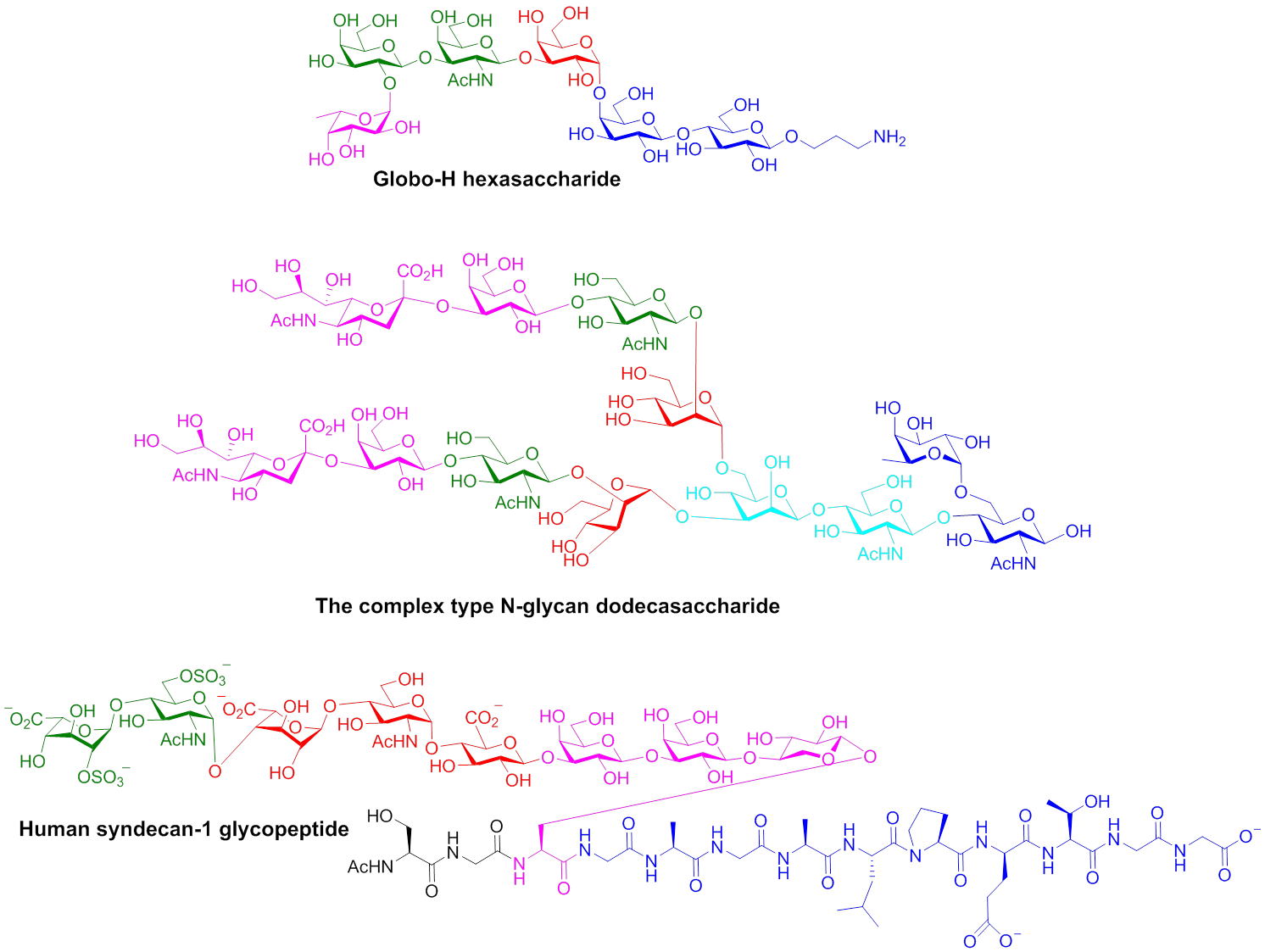
In the synthetic area, we are developing novel methodologies for assembling biologically active oligosaccharides and glycoconjugates. Traditional carbohydrate synthesis is very tedious and time-consuming. In order to expedite the synthetic process, we have developed novel one-pot glycosylation methodologies, where multiple sequential glycosylation reactions are carried out in a single reaction flask to yield desired oligosaccharides without time-consuming intermediate purifications. One of the methods we developed, the pre-activation based iterative one-pot method, has achieved higher synthetic efficiencies in several syntheses compared to the automated solid phase based method. We are applying the methods we developed to total synthesis of a wide range of highly complex oligosaccharides and glycoconjugates. Several representative examples of the molecules we have synthesized are shown below. We are continuing to synthesize biologically important carbohydrates.
Glyco-nanoscience
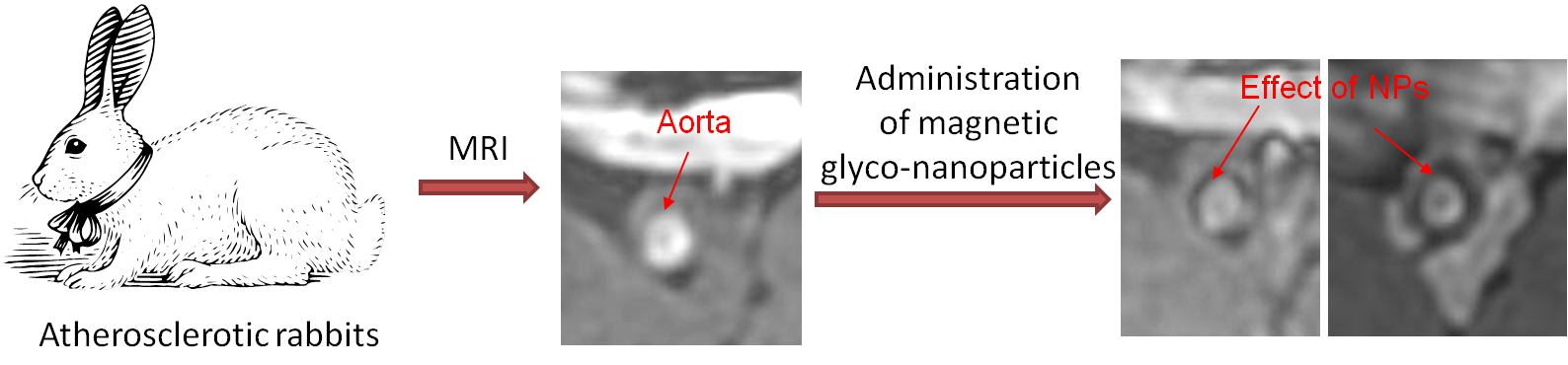
Carbohydrates play roles in many biological recognition events, which render them attractive as molecular recognition elements in biological sensor design. In our nanoscience program, we combine the multifaceted properties of carbohydrates with the unique functions of nanoparticles by immobilizing carbohydrates onto the external surface of magnetic nanoparticles. The magnetic glyco-nanoparticles (MGNPs) produced retain the biological recognition of carbohydrates and at the same time enhance the avidity of carbohydrate-receptor interactions by thousands of times. The magnetic nature of the MGNPs enables us to use magnetic resonance imaging (MRI) as a non-invasive method for disease detection. An example of this is shown below, where the presence of atherosclerotic plaques (the major cause of heart attack and stroke) in rabbits can be easily detected by MRI after injection of the MGNPs. Besides detection and imaging applications, we are exploring the utility of MGNPs for targeted drug delivery. We found that by incorporating drugs onto MGNPs, the cytotoxicity of the drugs towards cancer cells can be significantly enhanced. We are continuing to develop magnetic glyco-nanoparticles for non-invasive detection and treatment of diseases such as cancer, atherosclerosis and Alzheimer's disease.
Chemical Immunology
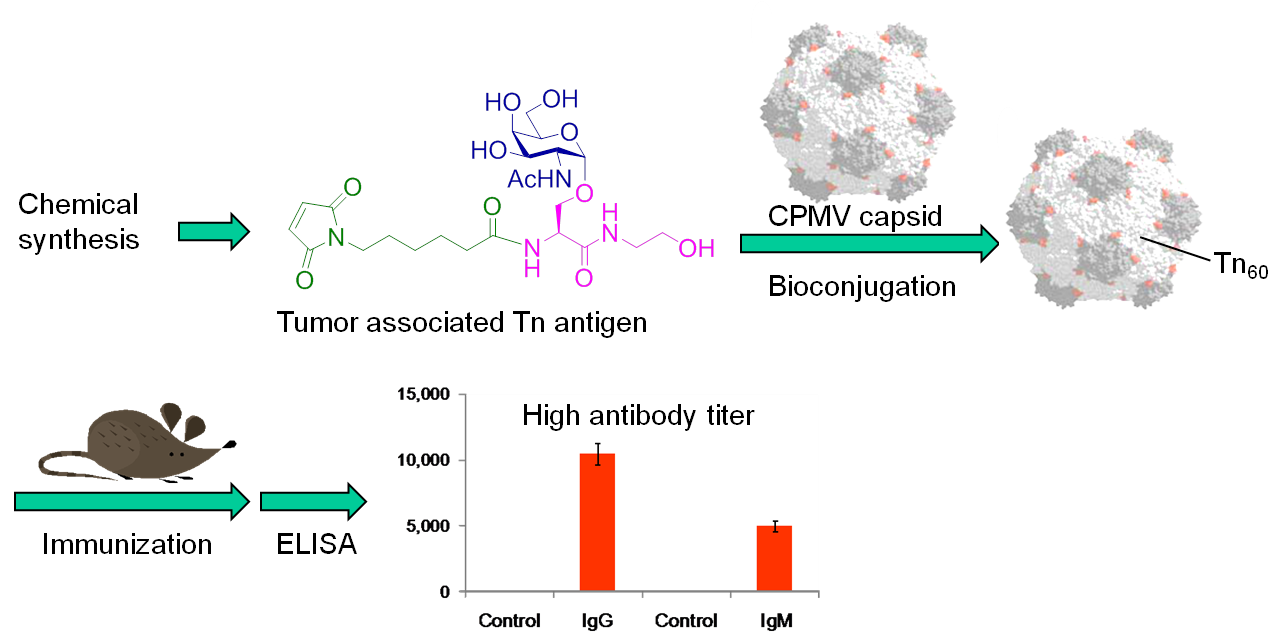
Harnessing the awesome power of body's immune system to fight cancer is an attractive strategy to cancer treatment. It is well known that many tumor cells have unique carbohydrate structures over-expressed on the cell surface. However, the low immunogenecities of these tumor associated carbohydrate antigens present a formidable challenge for the development of carbohydrate based anti-cancer vaccines. To overcome this obstacle, we are developing novel carrier systems such as cowpea mosaic virus capsid (CPMV) and bacteriophage Qβ to deliver tumor associated carbohydrate antigens to the immune system and to boost the immune responses against carbohydrates. We discovered that antigens displayed in a highly organized manner can elicit much stronger immune responses. Furthermore, the antibody generated can recognize antigens on the surfaces of cancer cells and lead to death of the cancer cells expressing the antigens. This is an excitingly new direction for the development of anti-cancer vaccines.